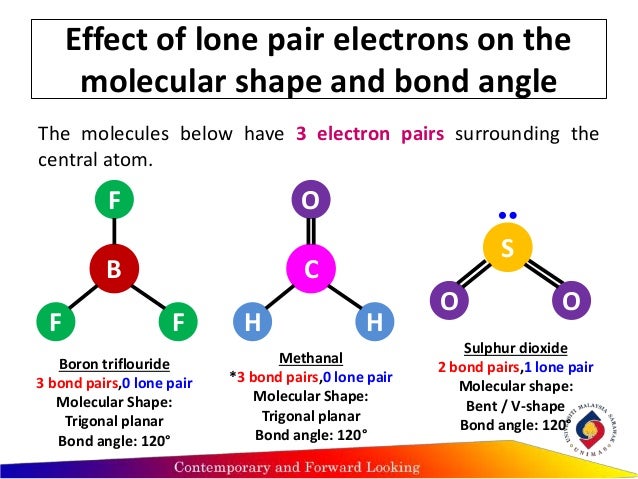 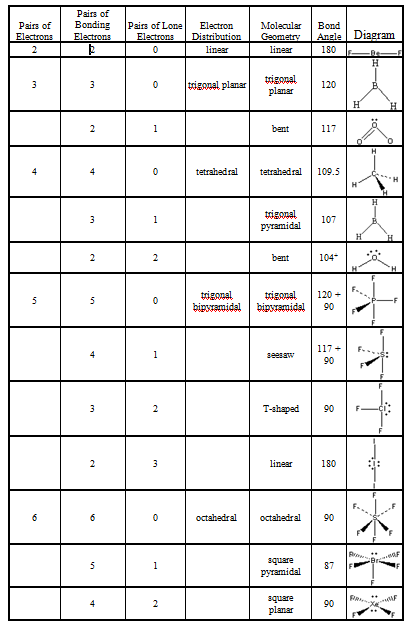 
Ammonia (NH 3) is not trigonal planar, however. For example, methane (CH 4) is tetrahedral-shaped because the carbon is attached to four hydrogens. The number of groups is how many atoms or electron pairs are bonded to the central atom. Those "things" can be other atoms or non-bonding pairs of electrons. The hybridization is determined by how many "things" are attached to the central atom. Table of Geometries Orbital Hybridization Modern methods of quantitatively calculating the most stable (lowest energy) shapes of molecules can take several hours of supercomputer time, and is the domain of computational chemistry. This can allow us to predict the geometry of similar molecules, making it a fairly useful model. The VSEPR model is by no means a perfect model of molecular shape! It is simply a system which explains the known shapes of molecular geometry as discovered by experiment. Wikipedia has related information at VSEPR Within this model, the AXE method is used in determining molecular geometry by counting the numbers of electrons and bonds related to the center atom(s) of the molecule. A scientific model, called the VSEPR (valence shell electron pair repulsion) model can be used to qualitatively predict the shapes of molecules. There are several different methods of determining molecular geometry. This repulsion causes covalent molecules to have distinctive shapes, known as the molecule's molecular geometry. Being mutually negatively charged, the electron pairs repel the other electron pairs and attempt to move as far apart as possible in order to stabilize the molecule. The electron-pair geometries will be the same as the molecular structures when there are no lone electron pairs around the central atom, but they will be different when there are lone pairs present on the central atom.Covalent molecules are bonded to other atoms by electron pairs. The structure that includes only the placement of the atoms in the molecule is called the molecular structure. We differentiate between these two situations by naming the geometry that includes all electron pairs the electron-pair geometry. Molecular structure describes the location of the atoms, not the electrons. The electron-pair geometries describe all regions where electrons are located, bonds as well as lone pairs. It is important to note that electron-pair geometry around a central atom is not the same thing as its molecular structure. The electrostatic repulsion of these electrons is reduced when the various regions of high electron density assume positions as far from each other as possible. The electrons in the valence shell of a central atom form either bonding pairs of electrons, located primarily between bonded atoms, or lone pairs. The VSEPR model assumes that electron pairs in the valence shell of a central atom will adopt an arrangement that minimizes repulsions between these electron pairs by maximizing the distance between them. Valence shell electron-pair repulsion theory (VSEPR theory) enables us to predict the molecular structure, including approximate bond angles around a central atom, of a molecule from an examination of the number of bonds and lone electron pairs in its Lewis structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
